Project Description
The goal of this project was to understand the formation of the universe and matter. This was done by a combination of models and math. We spent a good deal of time learning how chemical and nuclear reactions, fission, fusion, and nuclear decay, and the life of stars.
Things to know!
Subatomic particles:
Neutrons: Base particles of atoms with a charge of 0
Proton: Base particles of atoms with a charge of positive charge
Electron: Extremely small base particles with a negative charge.
Fission: When an unstable particle breaks apart into two, more stable atoms.
Fusion: When two atoms are pressed together hard enough, they can merge into a larger particle
Radioactive Decay: When an unstable atom breaks apart, and gives off radiation
Half-life: Unstable elements decay at semi regular rates. A half-life is how long it takes for half of a mass to decay
Nuclear Transmutation:
Radiation: The by products of radioactive decay
Nucleosynthesis: The formation of atoms larger than hydrogen.
Alpha Particle: A helium atom given off during radioactive decay
Beta Particle: An electron given off during radioactive decay
Gamma Rays: Energy given off during radioactive decay
Neutrons: Base particles of atoms with a charge of 0
Proton: Base particles of atoms with a charge of positive charge
Electron: Extremely small base particles with a negative charge.
Fission: When an unstable particle breaks apart into two, more stable atoms.
Fusion: When two atoms are pressed together hard enough, they can merge into a larger particle
Radioactive Decay: When an unstable atom breaks apart, and gives off radiation
Half-life: Unstable elements decay at semi regular rates. A half-life is how long it takes for half of a mass to decay
Nuclear Transmutation:
Radiation: The by products of radioactive decay
Nucleosynthesis: The formation of atoms larger than hydrogen.
Alpha Particle: A helium atom given off during radioactive decay
Beta Particle: An electron given off during radioactive decay
Gamma Rays: Energy given off during radioactive decay
Projects!
|
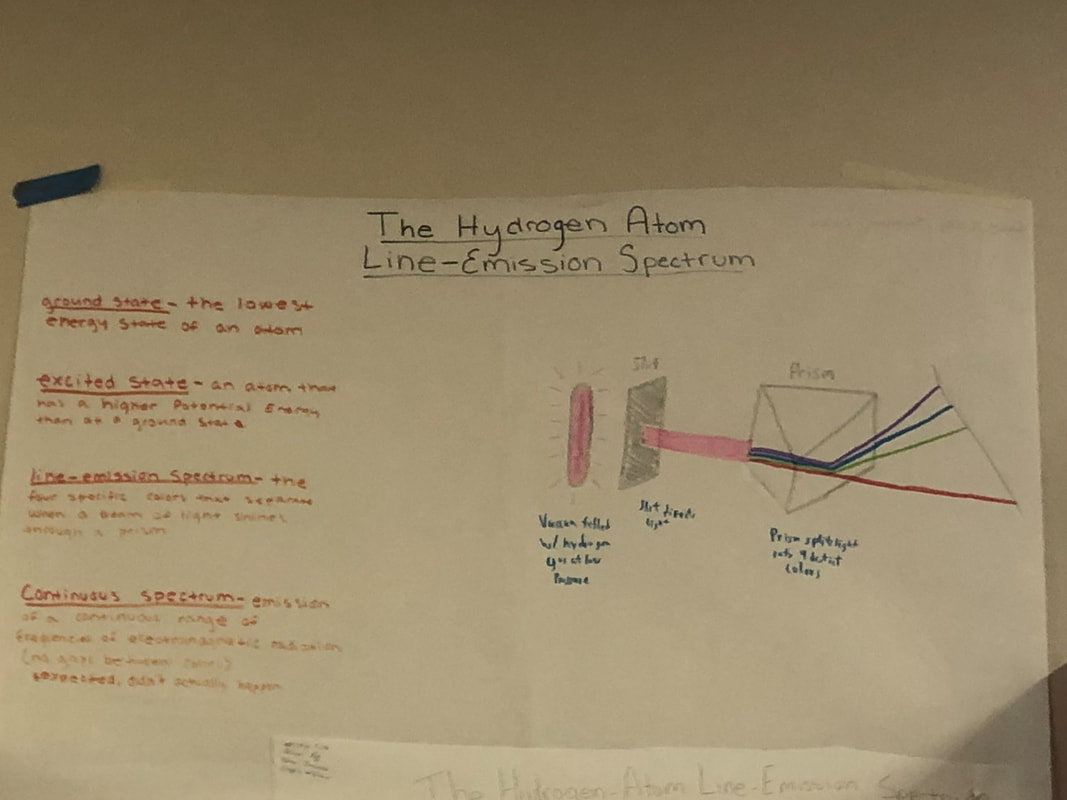
The second project was to learn and understand how light behaves when emitted. My group was tasked with explaining how light emitted from hydrogen emits light in very specific wave lengths. This is due to the fact that when hydrogen's electrons move down from an excited state, they emit light.
|
Reflection
I greatly enjoyed this project, however I feel I took a more reserved role, and didn't enjoy it as much as I should have, and rather than collaborating, simply took orders. I believe that if I had taken a more active role aside from research I could have also learned more about the topics. On the other hand, I greatly enjoyed the topics related to space and feel that I had a good understanding going in. To second this, I may have acted superior to my classmates who did not have the same work ethic or interest in the topics. I will work to resolve this in the future.